 
At the 3-month follow-up, he persisted with right upper extremity paresis, mild dysarthria, and speech apraxia, but without evidence of new episodes of hemolysis, thromboembolic events, or acute stroke. The patient was approved for initiation of immunotherapy with eculizumab 2 months after discharge. The patient was diagnosed with classical PNH associated with typical features of Moyamoya syndrome and referred to hematology for subsequent ambulatory follow-up. It detected CD59 deficiency in 50% of erythrocytes, CD16 and CD24 deficiency in 88% of neutrophils, CD24 deficiency in 94.5% of eosinophils, and CD14 deficiency in 91% of monocytes, consistent with defective clones immunophenotypically compatible with PNH. Serum chemistries and autoimmunity screening tests were normal.ĭue to the finding of hemolytic anemia in a patient presenting with ischemic stroke, having excluded immune and microangiopathic causes of hemolysis, flow cytometry/FLAER was performed. Haematologic findings included Coombs negative hemolytic anemia with a hemoglobin value of 6.6 g/dl, 4080 leukocytes/mm 3, 288 000 platelets/mm 3, a normal peripheral blood smear, elevated lactate dehydrogenase, increased indirect bilirubin, increased reticulocyte count, and decreased haptoglobin. Cerebrospinal fluid analysis was normal, as well as chest computerized tomography angiography and transthoracic echocardiography. Cerebral angiography showed multiple steno-occlusive changes in the proximal portion of both internal carotid arteries, with occlusion of the right M1 segment and significant stenosis at the origin of the left M1 segment, stenosis at the origin of the left P1 segment, and collateral flow arising from the right anterior cerebral artery (ACA) and supplying the territory corresponding to the posterior division of the right MCA, suggestive an intracranial arteriopathy ( Fig. He had a medical history of anemia, which had not been studied, and had once needed a red blood cell transfusion.īrain magnetic resonance angiography (MRA) evidenced a subacute ischemic stroke consistent with presenting symptoms, chronic bilateral ischemic events, and altered blood flow in MCAs. Due to the time between onset of symptoms and delayed arrival at our institution, he was not considered a candidate for fibrinolytic therapy. Case presentationĪ 41-year-old mestizo male patient was transferred to our hospital from another medical center, where he had been previously hospitalized for approximately 15 days because of acute right upper weakness, facial palsy, and dysarthria of sudden onset, without headache or other neurovegetative symptoms. We report the case of a young man with a medical history of Coombs-negative hemolytic anemia who had an ischemic stroke accompanied by steno-occlusive changes in intracranial arteries. 6 Moyamoya syndrome is rarely described in medical literature. 5 Involvement of intracranial and extracranial arterial sites is unusual, but when it occurs, middle cerebral arteries are the most commonly affected. 4 Arterial events, especially in coronary arteries, occur mostly in young patients. Brain involvement is rare and usually manifests as cerebral venous thrombosis or chronic small vessel disease, defined by silent periventricular and deep white matter lesions, multiple lacunar infarcts and/or microbleeds. Thrombotic events are the most serious complication associated with PNH and typically occur in unusual sites, such as dermal, mesenteric, hepatic, portal, splenic, and renal veins. These can be accompanied by hemoglobinuria, Coombs-negative hemolytic anemia, pulmonary hypertension, renal impairment, and, ultimately, death. 2Ĭlinical symptoms include weakness and fatigue on exertion, dyspnea, abdominal pain, and chest pain. However, GPI-deficient platelets and granulocytes are likely to be behind the thrombotic risk. 1 The affected erythrocytes are more susceptible to complement attack, triggering lysis. 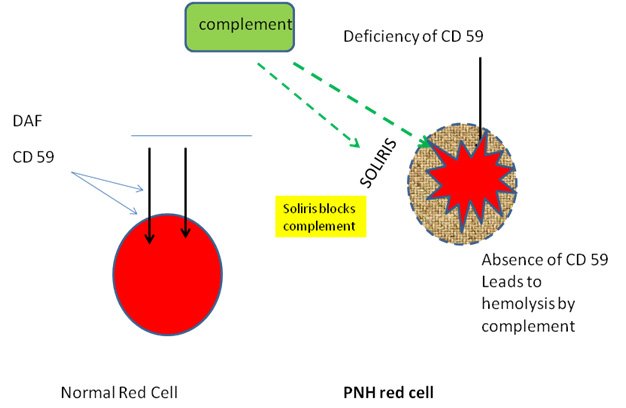
Clinical manifestations in PNH occur as a consequence of clonal expansion of haematopoietic stem cells deficient in GPI-anchored proteins due to somatic mutations in PIGA. This in turn can result in smooth muscle dystonias, thrombosis, and, in some cases, bone marrow failure. Paroxysmal nocturnal hemoglobinuria (PNH) is an infrequent, acquired disorder, in which impaired biosynthesis and defective surface expression in blood cells of various glycophosphatidylinositol (GPI)-anchored membrane proteins, such as complement inhibitors CD55 and CD59, can lead to complement-mediated hemolysis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
